PRESERVE 2: A phase 3, randomized, double-blind trial of trilaciclib versus placebo in patients receiving first- or second-line gemcitabine and carboplatin for locally advanced unresectable or metastatic triple-negative breast cancer
Shom Goel1; Joyce O’Shaughnessy2; Antoinette R. Tan3; Boris Krastev4; Hope Rugo5; Philippe Aftimos6; Denise A. Yardley7; Zoran Andric8; Curt Wolfgang9; Jessica A. Sorrentino9; Wenli Tao9; Andrew Beelen9; Rajesh Malik9; and Sarika Jain9
This presentation is the intellectual property of the author/presenter. Copies of this poster obtained through QR (Quick Response) and/or text key codes are for personal use only and may not be reproduced without written permission of the authors. Contact them at shom.goel@petermac.org or sjain@g1therapeutics.com for permission to reprint and/or distribute.
1 Peter MacCallum Cancer Centre, Melbourne, Australia; 2 Baylor University Medical Center, Texas Oncology, US Oncology, Dallas, TX, USA; 3 Levine Cancer Institute, Atrium Health, Charlotte, NC, USA; 4 MHAT Hospital for Women Health Nadezhda, Sofia, Bulgaria; 5 University of California San Francisco Comprehensive Cancer Center, San Francisco, CA, USA; 6 Institut Jules Bordet, Université Libre de Bruxelles, Brussels, Belgium; 7 Sarah Cannon Research Institute and Tennessee Oncology, Nashville, TN, USA; 8 Clinical Hospital Centre Bezanijska Kosa, Belgrade, Serbia; 9 G1 Therapeutics, Research Triangle Park, NC, USA
American Society of Clinical Oncology Annual Meeting | June 4–8, 2021 | Chicago, IL, USA
Introduction
- Chemotherapy, alone or in combination with immune checkpoint inhibitors, is the standard of care for patients with metastatic triple-negative breast cancer (mTNBC)1–3
- However, not all patients with programmed death-ligand 1 (PD-L1)–positive TNBC are appropriate candidates for immune checkpoint inhibitor treatment, and some patients with PD-L1–negative TNBC may not derive clinical benefit4
- Additionally, chemotherapy-induced immunosuppression may affect antitumor efficacy, owing to an inability of the host immune system to effectively mount a response against the cancer5
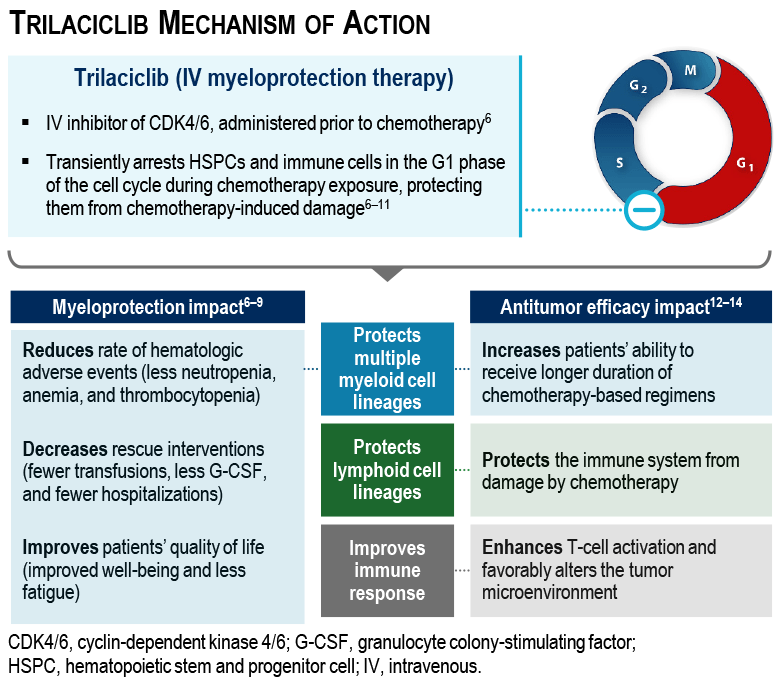
- Trilaciclib is an intravenous (IV) cyclin-dependent kinase (CDK)4/6 inhibitor indicated to decrease the incidence of chemotherapy-induced myelosuppression in adult patients when administered prior to a platinum/etoposide- or topotecan-containing chemotherapy regimen for extensive-stage small cell lung cancer6
- A randomized, open-label, phase 2 trial (NCT02978716) compared trilaciclib prior to gemcitabine plus carboplatin (GCb) with GCb alone in patients with TNBC13
- Although the primary endpoint of myeloprotection was not met, a clinically meaningful improvement in overall survival (OS) was observed in the intention-to-treat population with both PD-L1–positive and –negative tumors13,14
- Among all patients, median OS was 20.1 months with trilaciclib plus GCb versus 12.6 months with GCb alone (hazard ratio 0.36) in the primary analysis13
- Although the primary endpoint of myeloprotection was not met, a clinically meaningful improvement in overall survival (OS) was observed in the intention-to-treat population with both PD-L1–positive and –negative tumors13,14
Objectives
PRESERVE 2 STUDY
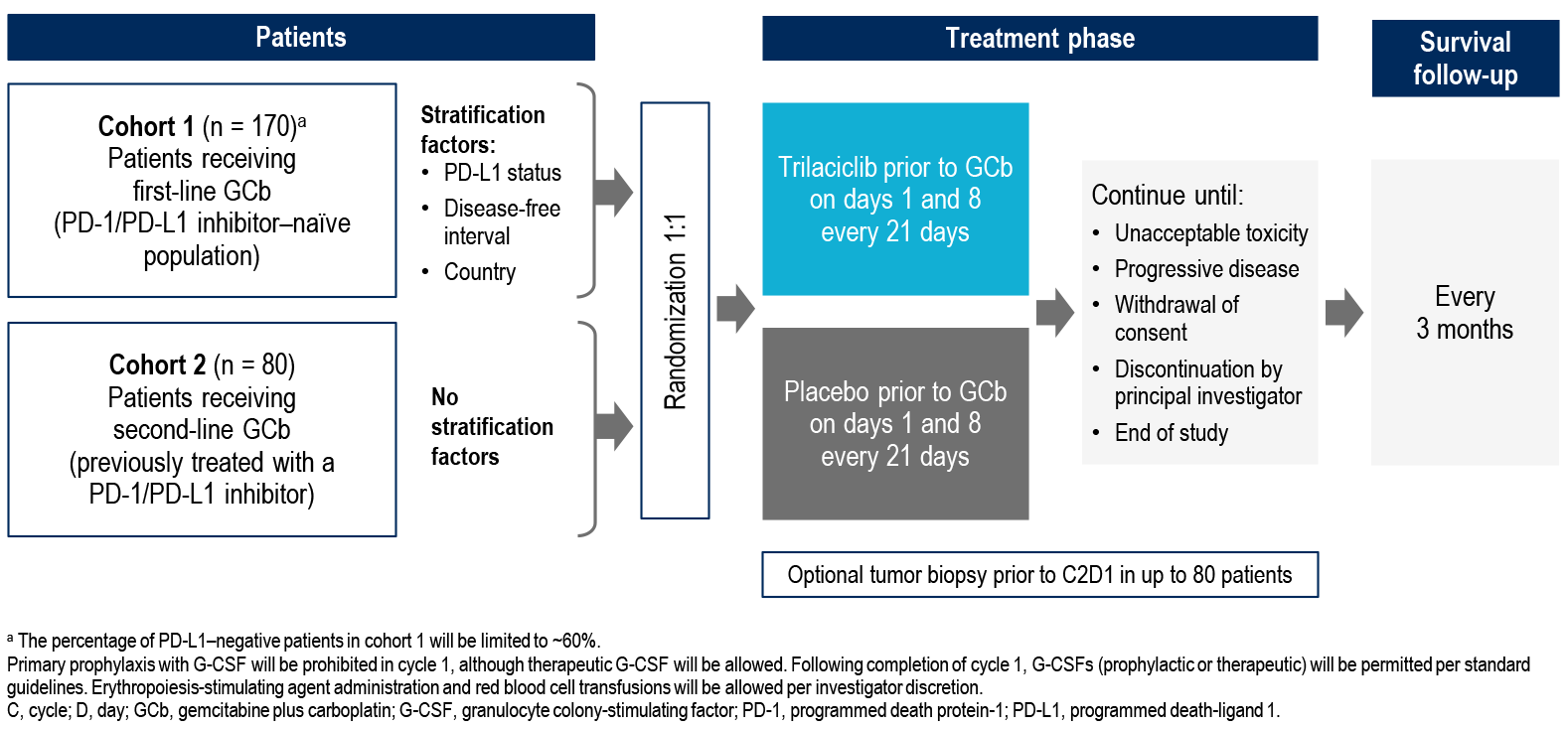
- PRESERVE 2 (NCT04799249) is a phase 3, multicenter, randomized, double-blind, placebo-controlled, 2-cohort study evaluating the safety and efficacy of trilaciclib versus placebo administered prior to GCb for patients receiving first- or second-line treatment for advanced/metastatic TNBC
- This study is designed to confirm the OS benefit seen in the phase 2 study, and to evaluate efficacy in a post–checkpoint inhibitor population
STUDY OBJECTIVES

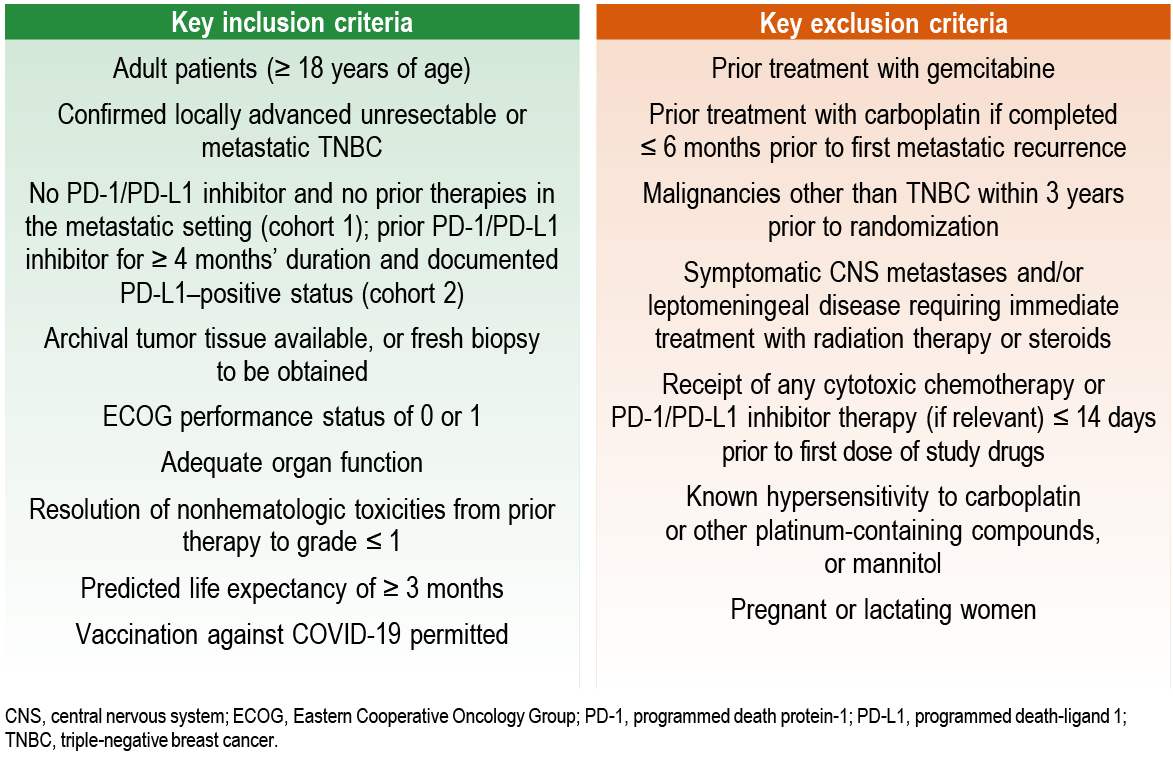
Patient Eligibility Criteria
Study Design
ENDPOINTS

OPTIONAL BIOPSY COLLECTION
- To evaluate the impact of trilaciclib on changes to the tumor-associated immune response in TNBC, immunophenotypic changes will be compared between tumor biopsies from patients receiving trilaciclib or placebo prior to GCb
- For patients who consent to optional biopsy collection, fresh tumor biopsies from a recurrent/metastatic lesion will be collected at baseline and on-treatment, prior to cycle 2
- Archival tissue is acceptable for use as the baseline sample if no systemic therapy or local radiation has been administered between biopsy and randomization
- Target participation for optional biopsy collection is 80 patients, including ~60 patients from cohort 1 and ~20 patients from cohort 2
STATISTICS
- Data from each cohort will be analyzed separately
- An interim analysis for OS will be performed for cohort 1 when ~70% of required events have been observed
- If the primary analysis of OS is statistically significant, then TTCD-fatigue will be analyzed
Study Sites
References and Acknowledgments
REFERENCES
- 1. Twelves C, et al. Crit Rev Oncol Hematol. 2016;100:74–87.
- 2. Cortes J, et al. Lancet. 2020;396:1817–28.
- 3. Schmid P, et al. N Engl J Med. 2018;379:2108–21.
- 4. Emens LA, et al. JAMA Oncol. 2019;5:74–82.
- 5. Zitvogel L, et al. Nat Rev Immunol. 2008;8:59–73.
- 6. COSELA™ (trilaciclib). Prescribing Information. https://www.g1therapeutics.com/cosela/pi/. Accessed April 2021
- 7. Daniel D, et al. Int J Cancer. 2021;148:2557–70.
- 8. Weiss JM, et al. Ann Oncol. 2019;30:1613–21.
- 9. Hart LL, et al. Adv Ther. 2021;38:350–65.
- 10. He S, et al. Sci Transl Med. 2017;9:eaal3986.
- 11. Li C, et al. Cancer Chemother Pharmacol. 2021;87:689–700.
- 12. Lai A, et al. J Immunother Cancer. 2020;8:e000847.
- 13. Tan AR, et al. Lancet Oncol. 2019;20:1587–601.
- 14. O’Shaughnessy J, et al. SABCS poster presentation. 2020; abstract #PD1-06.
ACKNOWLEDGMENTS
Study sponsored by G1 Therapeutics. Medical writing assistance was provided by Farhana Burnett, PhD, from Alligent Europe (Envision Pharma Group), funded by G1 Therapeutics.














