PRESERVE 1: A phase 3, randomized, double-blind trial of trilaciclib versus placebo in patients receiving FOLFOXIRI/bevacizumab for metastatic colorectal cancer
Joleen M. Hubbard1; Matti Aapro2; Satish Shah3; Afshin Eli Gabayan4; Richard Siegel5; Yili Pritchett6; John Yi6; Janet Horton6; and Fortunato Ciardiello7
For questions regarding the PRESERVE 1 trial, please contact clinicalinfo@g1therapeutics.com. Copies of this e-Poster obtained through QR, AR, and/or text key codes are for personal use only and may not be reproduced without written permission of the authors.
1Mayo Clinic, Rochester, MN, USA; 2Genolier Cancer Center, Clinique de Genolier, Genolier, Switzerland; 3Gettysburg Cancer Center, Gettysburg, PA, USA; 4Beverly Hills Cancer Center, Beverly Hills, CA, USA; 5Illinois Cancer Specialists, Arlington Heights, IL, USA; 6G1 Therapeutics, Inc., Research Triangle Park, NC, USA; 7University of Campania Luigi Vanvitelli, Naples, Italy
ESMO Congress 2021 | September 16–21, 2021 | Virtual
Introduction
- Multiagent chemotherapy remains the cornerstone of treatment for metastatic colorectal cancer (mCRC), with most patients receiving some combination of leucovorin, fluorouracil, oxaliplatin, and irinotecan in the first-line setting, often in combination with a monoclonal antibody targeting the epidermal growth factor receptor (EGFR) or vascular endothelial growth factor (VEGF) pathways1,2
-
Improvements in overall survival (OS) and progression-free survival (PFS) gained from combining leucovorin, fluorouracil, oxaliplatin, and irinotecan (FOLFOXIRI) with bevacizumab have come at the expense of increased chemotherapy-induced toxicity, including myelosuppression, diarrhea, and mucositis1-3
- As a result, the use of FOLFOXIRI is frequently limited to younger patients with fewer comorbidities
-
Chemotherapy-induced myelosuppression, which commonly manifests as neutropenia, anemia, and/or thrombocytopenia, is a dose-limiting and potentially fatal complication of treatment that can result in hospitalization and the need for supportive care interventions
- Symptoms of fatigue, and the development of infections and bleeding can seriously affect quality of life, and dose reductions and treatment delays may affect treatment response and long-term survival4,5
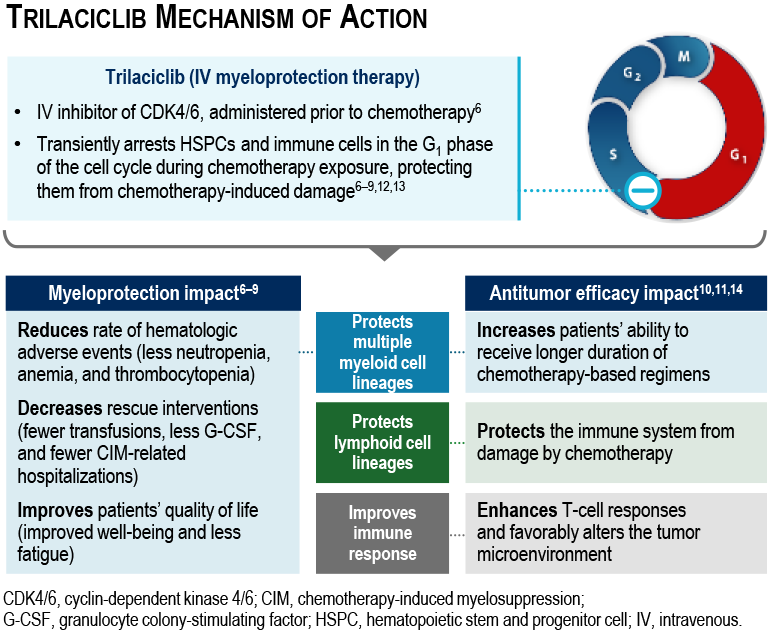
- Trilaciclib is an intravenous cyclin-dependent kinase 4/6 inhibitor indicated to decrease the incidence of chemotherapy-induced myelosuppression in adult patients when administered prior to a platinum/etoposide- or topotecan-containing chemotherapy regimen for extensive-stage small cell lung cancer6
- Data from 3 randomized, placebo-controlled, phase 2 clinical trials showed that administering trilaciclib prior to chemotherapy reduced the incidence of chemotherapy-induced myelosuppression, and reduced the need for supportive care interventions and chemotherapy dose reductions/delays7-9
- Additionally, in a randomized phase 2 trial in patients with metastatic triple-negative breast cancer, administering trilaciclib prior to gemcitabine plus carboplatin significantly improved OS compared with chemotherapy alone, potentially through protection and direct activation of immune function10,11
Objectives
PRESERVE 1 STUDY
- PRESERVE 1 (NCT04607668) is a phase 3, multicenter, randomized, double-blind, placebo-controlled study evaluating the myeloprotective and antitumor efficacy of trilaciclib versus placebo administered prior to FOLFOXIRI/bevacizumab for patients receiving first-line treatment for proficient mismatch repair/microsatellite stable (pMMR/MSS) mCRC
- FOLFOXIRI is more efficacious and more myelosuppressive than other fluorouracil-based regimens used in the treatment of mCRC; therefore, patients should benefit from a reduction in the incidence of chemotherapy-induced myelosuppression and use of this regimen at the standard-of-care dose and schedule
STUDY OBJECTIVES

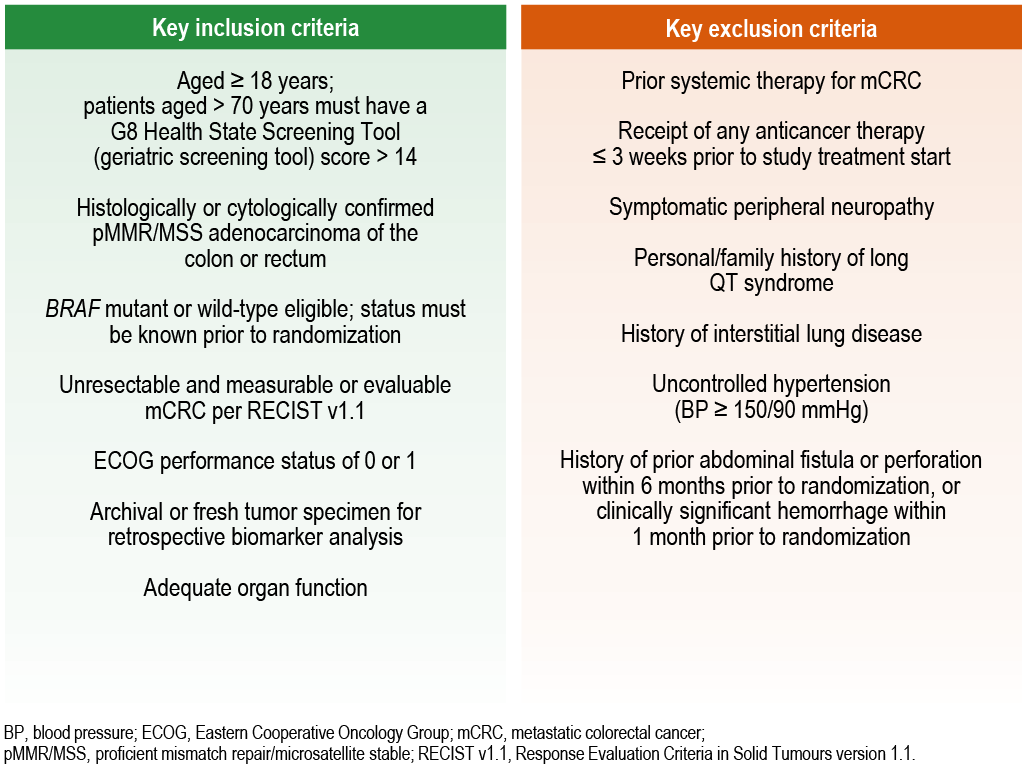
Patient Eligibility Criteria
Study Design
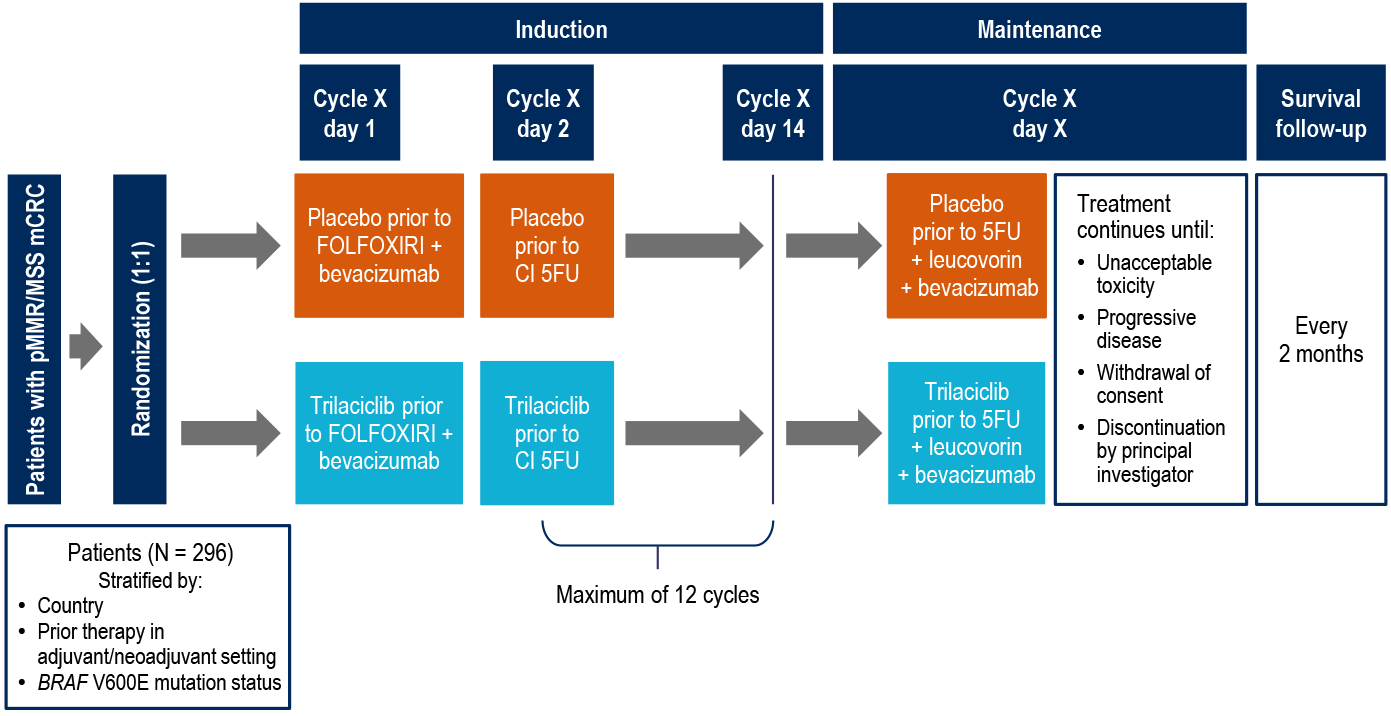
5FU, fluorouracil; CI, continuous infusion; FOLFOXIRI, leucovorin, fluorouracil, oxaliplatin, and irinotecan; mCRC, metastatic colorectal cancer; pMMR/MSS, proficient mismatch repair/microsatellite stable.
ENDPOINTS

STATISTICS
- To ensure strong control of family-wise type I error rate at the level of 2-sided 0.05, the following statistical considerations are specified in the protocol:
1. Overall alpha (α) will be split for analyses between 2 groups:- Group 1: analyses of the 2 primary myelosuppression endpoints and time to first confirmed deterioration in fatigue (TTCD-fatigue) using α1 = 0.04
- Group 2: analyses of PFS and OS using α2 = 0.01
2. Hierarchical procedures are specified to test treatment effects within each group:- As coprimary endpoints, duration of severe (grade 4) neutropenia (DSN) in cycle 1, and occurrence of severe neutropenia (SN) during induction will each be tested at the 2-sided 0.04 level; if both are positive, α1 will be passed to test treatment effect on TTCD-fatigue
- Treatment effect for OS will be tested after the treatment effect for PFS is established at the α2 level
3. Recycling of α from Group 1 to Group 2:- If the treatment effects are established for all 3 endpoints in Group 1, PFS and OS will be tested at the 2-sided 0.05 level; otherwise, they will be tested at the 0.01 level
- The sample size is determined to support the primary efficacy analysis for the 2 primary efficacy endpoints:
- 282 patients will be needed to detect treatment effect on DSN using a Mann-Whitney-Wilcoxon test, and on occurrence of SN using a chi-square test, with 90% power at the 2-sided significance level of 0.04
- Assuming 5% of randomized patients will not have any postbaseline data, a total of 296 patients (148 per group) will be required for the study
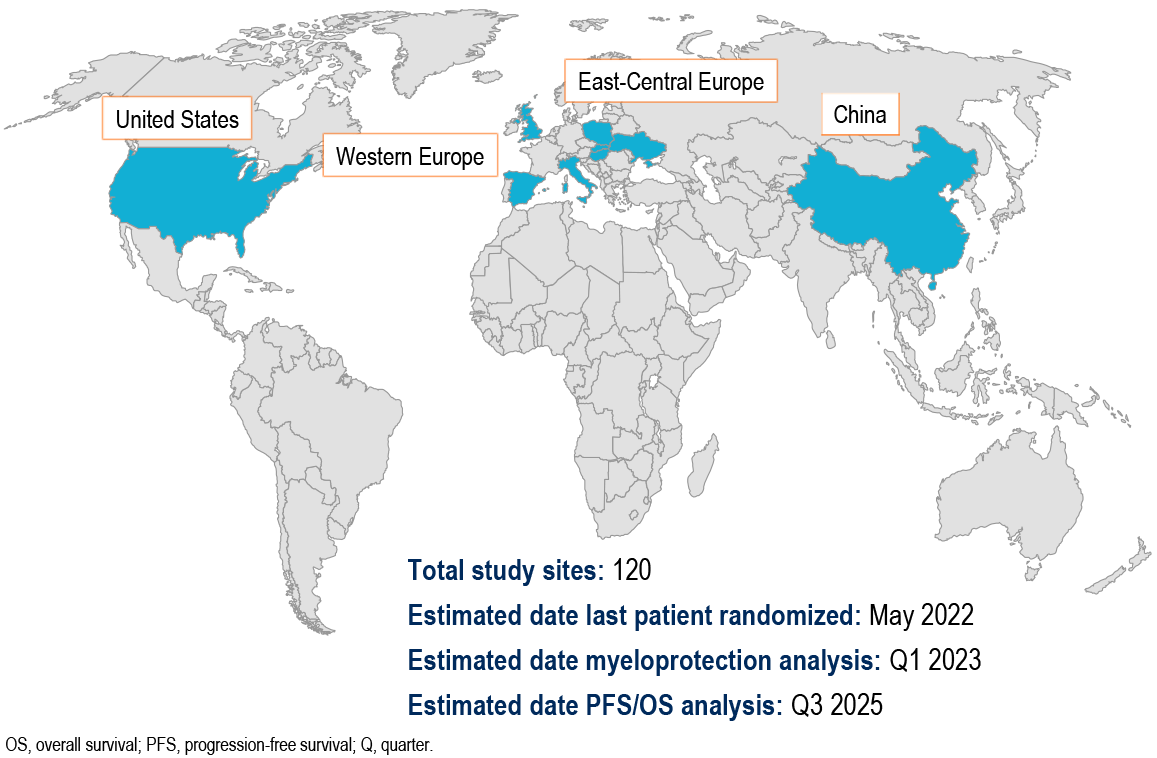
Study Sites
References and Acknowledgments
REFERENCES
- 1. Montagnani F, et al. Colorectal Dis. 2011;13:846–52.
- 2. Loupakis F, et al. N Engl J Med. 2014;371:1609–18.
- 3. Sastre J, et al. J Clin Oncol. 2019;37:3507
- 4. Epstein RS, et al. Adv Ther. 2020;37:3606–18.
- 5. Epstein RS, et al. Patient Prefer Adherence. 2021;15:453–65.
- 6. COSELA™ (trilaciclib). Prescribing Information. https://www.g1therapeutics.com/cosela/pi/. Accessed August 2021.
- 7. Daniel D, et al. Int J Cancer. 2021;148:2557–70.
- 8. Weiss JM, et al; G1T28-02 Study Group. Ann Oncol. 2019;30:1613–21.
- 9. Hart LL, et al. Adv Ther. 2021;38:350–65.
- 10. Tan AR, et al. Lancet Oncol. 2019;20:1587–601.
- 11. O’Shaughnessy J, et al. SABCS poster presentation. 2020; abstract #PD1-06.
- 12. He S, et al. Sci Transl Med. 2017;9:eaal3986.
- 13. Li C, et al. Cancer Chemother Pharmacol. 2021;87:689–700.
- 14. Lai AY, et al. J Immunother Cancer. 2020;8:e000847
ACKNOWLEDGMENTS
Study sponsored by G1 Therapeutics. Medical writing assistance was provided by Farhana Burnett, PhD, from Alligent Europe (Envision Pharma Group), funded by G1 Therapeutics, Inc.














